Researchers from Temasek Life Sciences Laboratory (TLL) and the Disruptive & Sustainable Technologies for Agricultural Precision (DiSTAP) Interdisciplinary Research Group (IRG) of Singapore-MIT Alliance for Research and Technology (SMART), the research arm of MIT in Singapore, have created the first ever nanosensors capable of detecting and differentiating gibberellins (GAs), a class of hormones in plants that are crucial for growth.
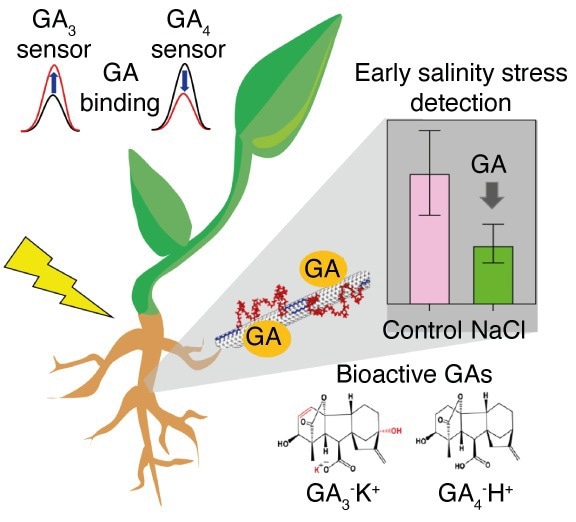
Illustration of GA detection in living plants using near-infrared fluorescent carbon nanotube sensors for early indication of salinity stress. Image Credit: SMART
Contrary to traditional collecting techniques, the innovative nanosensors have been successfully tested in living plants and are non-destructive.
The sensors could revolutionize agriculture and plant biotechnology when used in the field for early-stage plant stress monitoring, providing farmers interested in high-tech precision agriculture and crop management with a useful tool to maximize productivity.
The scientists created two plant hormones, GA3 and GA4, that can be detected and distinguished using near-infrared (NIR) fluorescent carbon nanotube sensors. GA3 and GA4 are diterpenoid phytohormones generated by plants that are members of the family of plant hormones known as gibberellins (GAs). They are crucial in influencing a variety of processes involved in plant growth and development.
GAs are believed to have contributed to the “green revolution” of the 1960s, which is credited for preventing starvation and saving countless lives throughout the world. Further research into gibberellins could result in new developments in agricultural science and have an impact on food security.
Farming soil becomes more salinized as a result of saltwater contamination brought on by climate change, global warming, and increasing sea levels. In turn, excessive soil salinity is known to favor GA metabolism and adversely affect GA production, which lowers the amount of GA in plants.
When used in the field, the new nanosensors created by the SMART researchers enable the investigation of GA dynamics in living plants under salinity stress at an extremely early stage, potentially enabling farmers to make early interventions. Early-stage stress detection is built on this.
Currently, GA3 and GA4 detection approaches use destructive and time-consuming mass spectroscopy-based analysis. The new sensors created by the researchers, on the other hand, are highly selective for the relevant GAs and provide in vivo, real-time monitoring of changes in GA levels across a variety of plant species.
The study, which was detailed in a paper titled “Near-Infrared Fluorescent Carbon Nanotube Sensors for the Plant Hormone Family Gibberellins” and published in the journal Nano Letters, represents a significant breakthrough in the early detection of plant stress and has the potential to significantly advance plant biotechnology and agriculture.
This publication extends earlier work on SWNT-based nanosensors conducted by the SMART DiSTAP team utilizing the corona phase molecular recognition (CoPhMoRe) platform.
The novel sensors, which are based on the CoPhMoRe idea developed by the Strano Lab at MIT, are capable of detecting GA kinetics in the roots of several model and non-model plant species, such as Arabidopsis, lettuce, and basil, in addition to GA accumulation during lateral root emergence, highlighting the significance of GA in root system architecture.
This was made possible by the researchers’ related work on a new coupled Raman/NIR fluorimeter that allows for the self-referencing of nanosensor NIR fluorescence with its Raman G-band.
This new hardware innovation eliminates the need for a separate reference nanosensor and cuts down the instrumentation necessities by using a single optical channel to measure hormone concentration.
The researchers discovered lower endogenous GA levels in plants under salinity stress and higher endogenous GA levels in mutant plants that produced more of the essential GA biosynthesis enzyme GA20ox1.
Researchers also discovered that lettuce’s development was significantly reduced when subjected to salinity stress; this effect only became apparent after 10 days. The GA nanosensors, on the other hand, indicated lower GA levels after just 6 hours, proving their effectiveness as a considerably early indicator of salinity stress.
Our CoPhMoRe technique allows us to create nanoparticles that act like natural antibodies in that they can recognize and lock onto specific molecules. But they tend to be far more stable than alternatives. We have used this method to successfully create nanosensors for plant signals such as hydrogen peroxide and heavy-metal pollutants like arsenic in plants and soil.
Michael Strano, Study Corresponding Author, DiSTAP Co-Lead Principal Investigator and Carbon P. Dubbs Professor, Chemical Engineering, Massachusetts Institute of Technology
Strano added, “The method works to create sensors for organic molecules like synthetic auxin—an important plant hormone—as we have shown. This latest breakthrough now extends this success to a plant hormone family called gibberellins—an exceedingly difficult one to recognize.”
“The resulting technology offers a rapid, real-time, and in vivo method to monitor changes in GA levels in virtually any plant, and can replace current sensing methods which are laborious, destructive, species-specific and much less efficient,” Strano further added.
Dr Mervin Chun-Yi Ang, Associate Scientific Director at DiSTAP and the study’s co-first author, further stated, “More than simply a breakthrough in plant stress detection, we have also demonstrated a hardware innovation in the form of a new coupled Raman/NIR fluorimeter that enabled self-referencing of SWNT sensor fluorescence with its Raman G-band, representing a major advance in the translation of our nanosensing toolsets to the field.”
He continued, “In the near future, our sensors can be combined with low-cost electronics, portable optodes, or microneedle interfaces for industrial use, transforming how the industry screens for and mitigates plant stress in food crops and potentially improving growth and yield.”
The new sensors could still be used in a wide range of industrial applications.
GAs are known to regulate a wide range of plant development processes, from shoot, root, and flower development, to seed germination and plant stress responses. With the commercialization of GAs, these plant hormones are also sold to growers and farmers as plant growth regulators to promote plant growth and seed germination. Our novel GA nanosensors could be applied in the field for early-stage plant stress monitoring, and also be used by growers and farmers to track the uptake or metabolism of GA in their crops.
Daisuke Urano, Study Co-Corresponding Author and Adjunct Assistant Professor, National University of Singapore
While TLL was in charge of the design, execution, and analysis of plant-related studies, including the validation of nanosensors in living plants, SMART and MIT designed and developed the nanosensors, created and validated the coupled Raman/NIR fluorimeter and associated image/data processing algorithms, and also statistically analyzed readouts from plant sensors for this study.
The study is being conducted by SMART with funding from NRF as part of its Campus for Research Excellence and Technological Enterprise (CREATE) initiative.
Journal Reference
Boonyaves, K., et al. (2023) Near-Infrared Fluorescent Carbon Nanotube Sensors for the Plant Hormone Family Gibberellins. Nano Letters. doi:10.1021/acs.nanolett.2c04128.